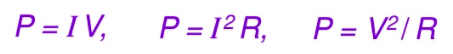|
This is from the official IGCSE website, it will help you link the different chapters in order to be able to answer questions that involve the related chapters! You can use this for revision and make sure you understand the connections between the topics.
0 Comments
Unit Three - Waves and Sounds
There are two types of waves:
Rarefactions - Areas of low pressure where the molecules are farther apart. Important Definitions -
The wave equation - SPEED = FREQUENCY x WAVELENGTH Wave Effects
So the - SPEED DECREASES WAVELENGTH DECREASES FREQUENCY ALWAYS REMAINS CONSTANT !! IWhen waves travel from SHALLOW to DEEP water, the waves travel from a DENSER to a RARER medium, thus the ray bends AWAY FROM THE NORMAL. . So the- SPEED INCREASES WAVELENGTH INCREASES FREQUENCY ALWAYS REMAINS CONSTANT !!
If the gap is comparable to the wavelength then diffraction is significant and takes place. If the gap is lesser than the wavelength, greater diffraction occurs. Keyword to describe diffraction - angular spreading Note - Sound Waves cannot travel through vacuum. Speed of sound = Distance traveled / Time taken For echoes’ speed = 2*Distance traveled / Time taken FREQUENCY affects the PITCH of the sound Higher the frequency, higher the pitch (when the number of waves increase) AMPLITUDE affects the LOUDNESS of the sound Higher the amplitude, higher the loudness (longer distance between the X axis and the topmost part of the wave) Magnetism Properties of Magnets:
Magnetic Fields: The pattern of the magnetic field lines can be shown by sprinkling iron filings around a bar magnet. The filings arrange themselves in a way to show the magnetic field lines. The magnetic field lines and their directions can also be plotted using a compass. Remember: The field lines never cross over or touch each other, and are always drawn from the north pole to the south pole. Magnetic materials are called Ferrous. Magnets can only be tested by repulsion, because only two magnets will repel each other, while magnets can attract unmagnetised magnetic objects. Methods of Magnetisation:
Soft magnetic materials are easy to magnetise, but they also lose their magnetism easily. Example: Iron. Charges
--- When we rub two materials together, no electric charge is created instead it separates the charges that are already present.
Electrostatic force can be detected using a gold leaf electroscope
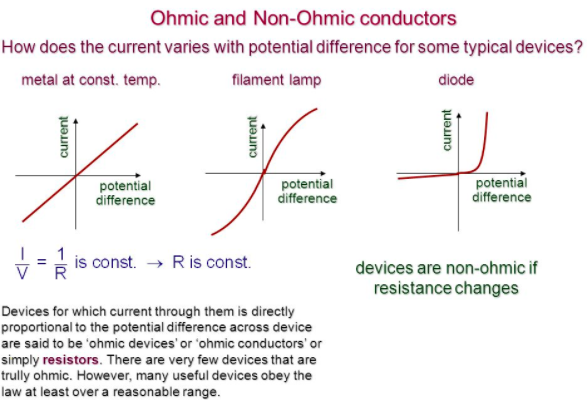
Factors that affect resistance
Learn the shapes of the graphs above in order to understand ohm’s law Ohm’s law states that the current of a component is directly proportional to the potential flowing across it provided temperature remains CONSTANT. Using the above formula you can find out the length/area or resistance of a wire of the same material of another wire where one of these variables is unknown.
(connecting this to the equation we learnt in the previous units also helps us connect energy to these electrical quantities! Just substitute P= energy/time ) Electric Circuits
Dangers of electricity Potential Hazards:
Safety Measures:
The magnetic effect of current
The direction of the magnetic field can be found by the right-hand grip rule. - If the thumb of your right hand points in the same direction as the current, then your fingers curl around the wire in the direction of the magnetic field. A solenoid is usually used to produce a magnetic field. It has the following features.
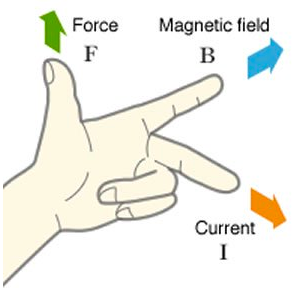
Force on a current- carrying conductor When a current carrying conductor is placed in a magnetic field, it will experience a force, and this force causes the conductor to move if it is free to do so. This is because the current carrying wire has its own magnetic field which interacts with the field of the permanent magnet. If the current carrying wire is placed in a magnetic field whose lines of force are at right angles to the wire, then it will experience a force at right angles to the current direction and the magnetic field lines. If the direction of the current or magnetic field is changed then the direction of the force also changes. But if both the direction of the current and the magnetic field are reversed, there is no change. This effect is used in motors, and can be increased by:
To remember:
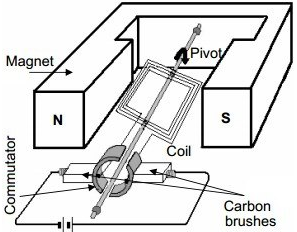
D.C. Motor
Working :
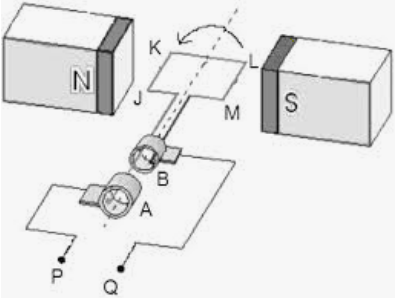
Electromagnetic Induction Electricity can be generated from a magnetic field using electromagnetic induction. Electromagnetic force (e.m.f.) is induced when a wire is moved through a magnetic field as the wire cuts the magnetic field. Induced e.m.f. Can be increased by:
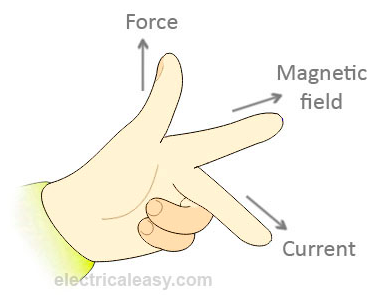
Remember: the wire must cut magnetic field lines for an e.m.f. to be induced. No e.m.f. will be induced if the wire is moved parallel to the magnetic field. Fleming’s left hand rule can be used to indicate the direction of a current caused by an induced e.m.f. . To remember:
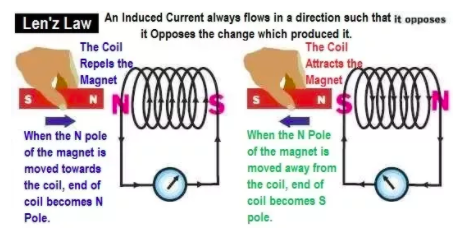
Induced current in a coil: Lenz’s law: The direction of the current opposes the change causing it. Induced e.m.f. can be increased by:
A.C. Generator A.C. Generator produces alternating e.m.f. current. It consists of a rotating coil between fixed magnets, and each end of the coil is connected to a slip ring against which the carbon brushes pressed. Working:
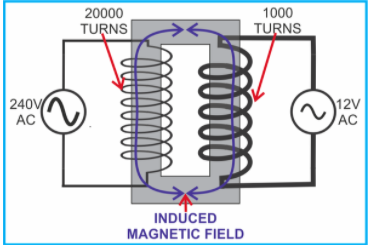
It is used to change an alternating voltage from one size into another. It consists of two insulated coils wound around a soft iron core. These are called primary and secondary coils.
vp= primary voltage. vs= secondary voltage. np= number of turns in primary coil. ns= number of turns in secondary coil. LIGHT
Monochromatic Light - light of a single wavelength, and color Real Image - An image that can be formed on a screen Virtual Image - An image that cannot be formed on a screen Refractive Index - Speed of light in vacuum / Speed of light in medium Refractive Index (alternate) - sin i / sin r Sin C = 1/n C = critical angle n = refractive index Refraction by a prism - The splitting into the range of colors is known as dispersion. Critical Angle The angle of incidence when the angle of refraction is 90 degrees. Total Internal Reflection
USES OF TOTAL INTERNAL REFLECTION
Convex Lenses Principle Focus - The point where the rays converge. Focal Length - The distance from the centre of the lens is the focal length. If the object is BEYOND 2F The image will be - REAL, INVERTED AND DIMINISHED If the object is BETWEEN 2F AND F The image will be - REAL, INVERTED AND ENLARGED If the object is BETWEEN F AND THE LENS The image will be - VIRTUAL, UPRIGHT AND MAGNIFIED ELECTROMAGNETIC SPECTRUM
This is one of the simpler units, so make sure you nail this :)
Atomic Model: Structure and properties of the nucleus:
The nuclear structure of the atom can be proved by Rutherford’s Gold Foil experiment.
Nuclear Fission: An atom usually undergoes radioactive decay. However, sometimes atoms such as Uranium-235 can break up in one instant. For this, it requires energy which can be provided by hitting the nucleus with a neutron. Two ‘daughter’ nuclei and two to three neutrons are produced. Kinetic energy is released in this process. Nuclear Fusion: When smaller nuclei are made to collide with each other at high speeds, they fuse to form larger nuclei. Energy is released in this process. Detection of Radiation: Background radiation: There is a small amount of radiation present around us all the time because of radioactive materials in the environment. This radiation is called background radiation and comes from both natural and man-made sources. Alpha radiation: The unstable parent nucleus decays into a stable nucleus by emitting alpha particles. The proton number reduces by 2 and the mass number reduces by 4. It can be absorbed by a few sheets of paper. Alpha radiation is rather dangerous inside the body. It is absorbed by the cells and it ionises them. It can cause eye cataracts, cancer, skin burns, leukaemia and other blood disorders. Beta radiation: The neutron in the parent nucleus changes to a proton and an electron. The electron escapes in the form of a beta particle and the proton joins with other protons in the nucleus. It can be absorbed by a thin sheet of metal such as aluminium. Gamma radiation: The gamma radiation represents simple loss of energy from the nucleus. The gamma rays are released when an atomic nucleus releases its excess energy. Emission of gamma rays does not change the mass number or atomic number. A thick sheet of lead or iron can absorb it. Beta and Gamma radiation are the most dangerous. They can penetrate and reach the cells of organs. They can cause cancer. In electric fields, Alpha particles move towards the negative plate and Beta particles move towards the positive plate. Beta particles are deflected more than Alpha particles. Radioactive decay is random. Half-Life: All the radioactive substances decay in an exponential pattern. Initially, the slope is steep. It is initially decaying very fast. The half-life of an isotope is the time taken for the activity of that isotope [or the number of original nuclei] to drop to half of its original value The number of half-life’s passed = Time elapsed / Half Life of the isotope. When calculating the half-life, remember to subtract the background radiation from the first reading, before dividing it by two and using the above formula! Activity: It is the number of atoms that decay each second and is measured in Bq [Becquerels] and the activity of one Bq is one decay per second. The count rate and activity both decrease following the same pattern as the number of undecayed atoms. Safety Precautions:
Physics Formulas
The number of significant figures needed in the answer has been a source of difficulty and change over the years. You will find that data given in the questions are usually given to two significant figures and the answers quoted in the marking scheme are also given to two sig. figs. Recently we have been penalising the use of more than 2 or 3 significant figures once in a question, so that a student writing down, say, four significant figures might lose one mark per question.
However this can mean that a student might lose a considerable number os marks overall in the paper and this policy has been relaxed. There is also the problem that when a student tries to write down the answer to 2 sig. figs. he or she might round wrongly writing, say, 1.67 as 1.6 rather than 1.7. Now we are allowing students in the theory paper to write down as many sig. figs as they like as long as it is more than just one (so 2 in the answer above is not accepted but 1.6666 would be. There is still the possibility that a question might ask for the answer to be given to a reasonable number of sig. figs when that would be expected to be the smallest number given in the question. The practical papers might be different because one is measuring to a certain accuracy given by the instrument being used, such as a ruler when one expects the answer to that accuracy, usually 1 mm. and so a value of 34.6 cm would be expected. Calculations after that usually mean that the answer of the calculation should be to the least number of significant figures. One also expects all the answers down a column of readings to have the same number of sig. figs.; thus 2.3 cm , 4.5 cm and 6 cm would be penalised because the 6 should be 6.0. Many of my own students used to lose a mark for forgetting the 0. I used to ask them to check all the readings were to the same precision. The mark scheme shows the answer to 2 sig. figs. and for some time now we accept more sig. figs and certainly 0.0228 J would be OK but not 0.02 J. When you add or subtract numbers known to 2 sig. figs. then the answer is effectively also know to 2 sig. figs. Again when you multiply or divide we just expect the number of significant figures in the result to be the least number of significant figures in the data provided. It is is a simple rule of thumb which uses the idea that if one quotes say a force as 0.67 N and a distance as 0.034 m then one is implying there is an uncertainty of 0.005 N in the force and 0.005 m in the distance (because otherwise if the force was 0.0676 you would write down 0.68 N). If you work out the maximum work 0.675 x 0.0345 = 0.02329 and you can see the first 2 sig. figs (2 and 3) are correct but the third is not as 0.67 x 0.034 = 0.02278. A sensitive thermometer is one where there is a large change in the reading for a small change in temperature. To compare liquid-in-glass thermometers, the thread in a sensitive thermometer will move further up the scale than in a less sensitive thermometer. Sensitive thermometers have a thin bore so that the liquid moves further up the tube for any expansion, more liquid to produce a larger change in volume during expansion and use a liquid that has a high expansivity (although this word is not in the course).
The common misunderstanding shown by many students is that they wrongly state that a sensitive thermometer is one that takes a short time to reach the correct temperature and that this is related to the thickness of the glass in the bulb. A thin glass bulb does cause the thermometer to reach the final reading faster but this is not what is meant by sensitivity. sensitivity can be assigned a unit, for a liquid-in-glass thermometer it is the number of mm that the thread moves up the scale for each °C change in temperature, i.e mm/°C. Other thermometers gave different units, for a thermocouple thermometer the unit is V / °C or, more likely, μV / °C. linearity can also be assigned a unit because the number of mm/°C at a low temperature is the same as at a higher temperature.
|
|||||||||||||